 |
| Science Panda & Science Zebra |
We then continued with mixing a solution of bicarbonate of soda and citric acid, making a 'fizzing frenzy'.
 |
| Fizzing frenzy. |
There is a brief explanation in the instruction booklet, which explains that this is a chemical reaction and that the bubbles and fizz are caused by the carbon dioxide escaping from the liquid. M wasn't particularly interested, but K & I went on to talk more about chemical reactions and compared them to algebraic equations. With a bit of googling, I found this, which included this chemical reaction:
H3C6H5O7(aq) + 3 NaHCO3(s) → 3 CO2(g) + 3 H2O(l) + Na3C6H5O7(aq)
We counted up how many atoms of each of hydrogen, carbon, oxygen and sodium there are on each side of the reaction. We also talked about atoms and molecules, using the example of water, with two hydrogen atoms and one oxygen atom to make one water molecule like this.
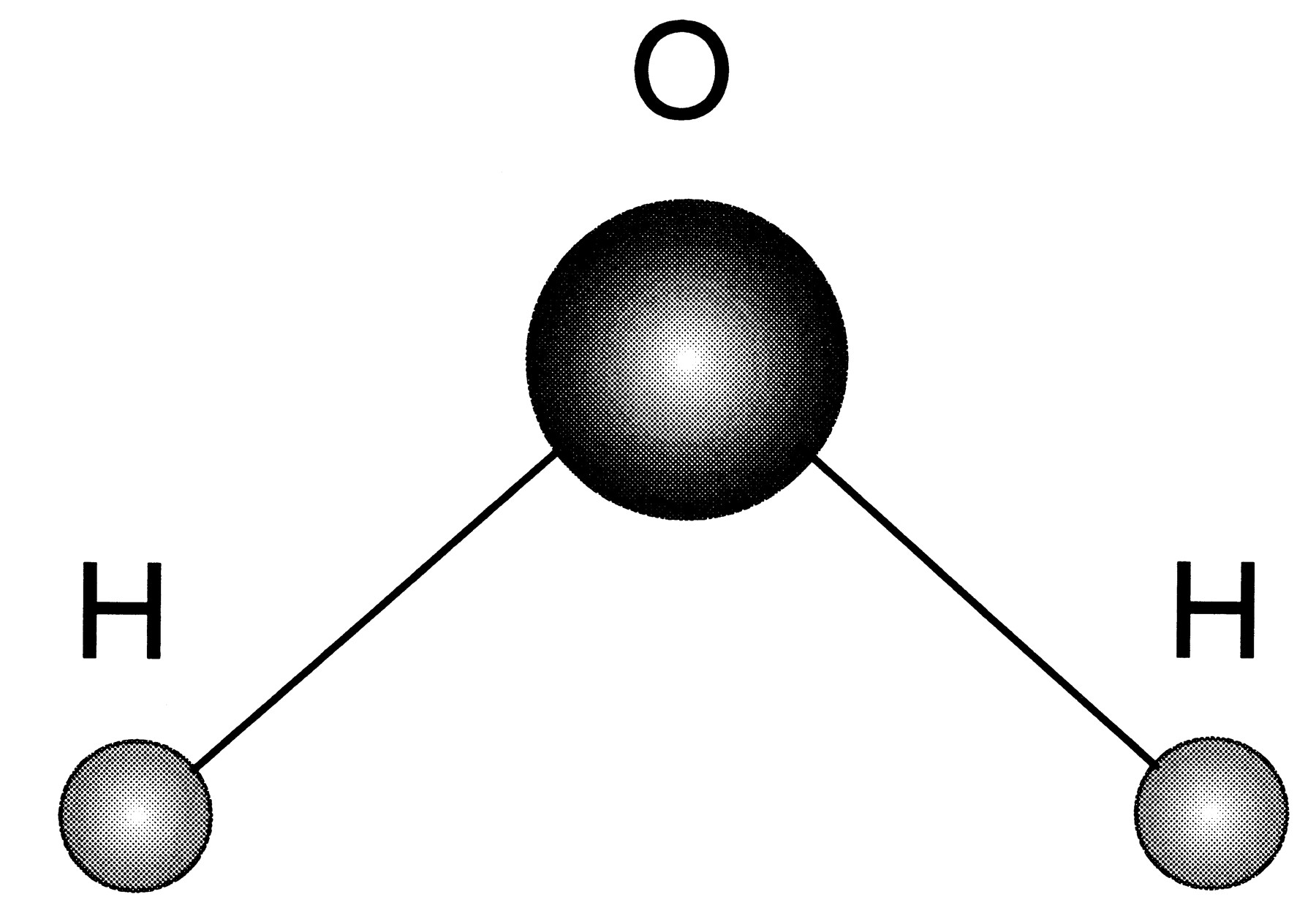 |
| A water molecule. |
M rejoined us to carry on with the experiments in the booklet, the next one being 'Hocus Pocus Powder', which involved using red cabbage juice powder as an indicator. K & M disolved the powder in water and saw that it turned purple. Then they added some citric acid to one test tube and watched the purple liquid turn red and some bicarb to another, which turned it blue.
 |
I then added a bit of a mystery liquid to some more indicator and asked the girls if they thought it was acid or alkaline, they immediately identified it as an acid. It was white vinegar.
The final experiment for today was basically a combination of the Hocus Pocus Powder and Fizzing Frenzy. Unfortunately the 'magic potion that will blow your mind'; combining the indicator with bicarb with that with citric acid was a complete damp squib, so we added a bit more of each of the citric acid and bicard to the test tube and got a bit more of a fizz and then when M added even more bicarb it fizzed rather impressively!
 |
In addition to all the 'Magic Science', we had friends come round for a while this afternoon for a cuppa and to help finish off the last of my birthday cake. K, M & I also gave them a rendition of The School School from Matilda the Musical.
I also started reading a new book to the girls, White Boots by Noel Streatfeild, so all in all a very productive day!
No comments:
Post a Comment